It is now recognized that emission CO2 is a major contributor to climate change. Combustion of fossil fuels is a major source of CO2 and serious efforts to mitigate these emissions are now being explored. Nature has a powerful method for abatement of carbon dioxide – it is called photosynthesis. In photosynthesis, the plant uses light to convert water and carbon dioxide into carbohydrates. It may be possible to mimic such a process using inorganic materials to give so-called artificial photosynthesis. The problems associated with developing such a nanocatalyst are challenging. The material must be effective at light harvesting in the visible region of the spectrum, carbon dioxide must be activated on the surface of the nanocatalyst and then brought together activated water to create a useful fuel. Such a system may not be realizable for many years but we are undertaking fundamental research to develop components of such a catalyst.
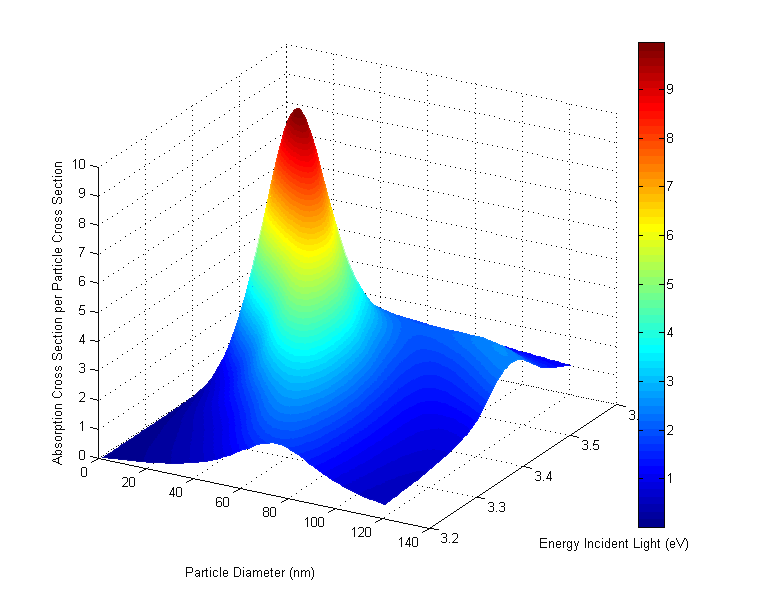
Calculations of the light harvesting capability of a Ag nanoparticles. The graph plots light absorption as a function of particles size and the energy of the light. Light absorption is strongest for 8nm particles and photon energy about 3.5 eV (in ultraviolet region of spectrum).